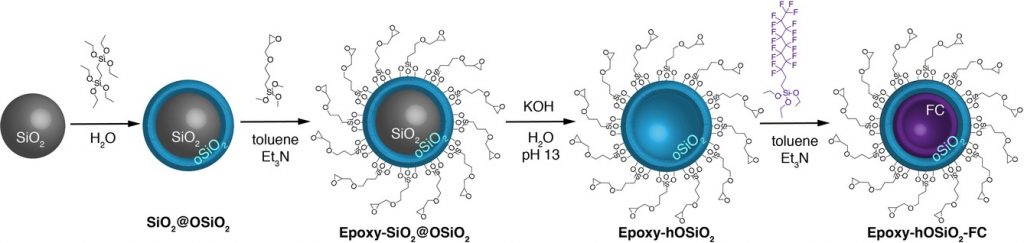
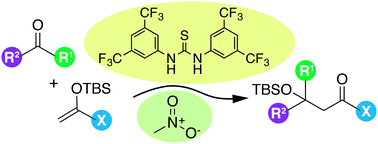
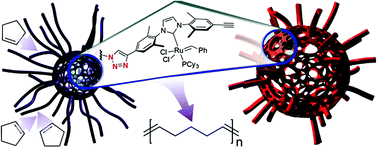
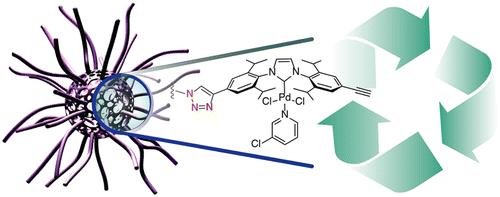
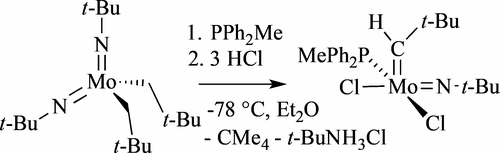FIU:
44. L. Wang, G. Castro, O. Alkhamis, M. R. Gangireddy, K. V. Bukhryakov, N. R. Vinueza, and Y. Xiao,* A Selective, Class-Specific Aptamer for Fentanyl Analog Screening, 2026, submitted.
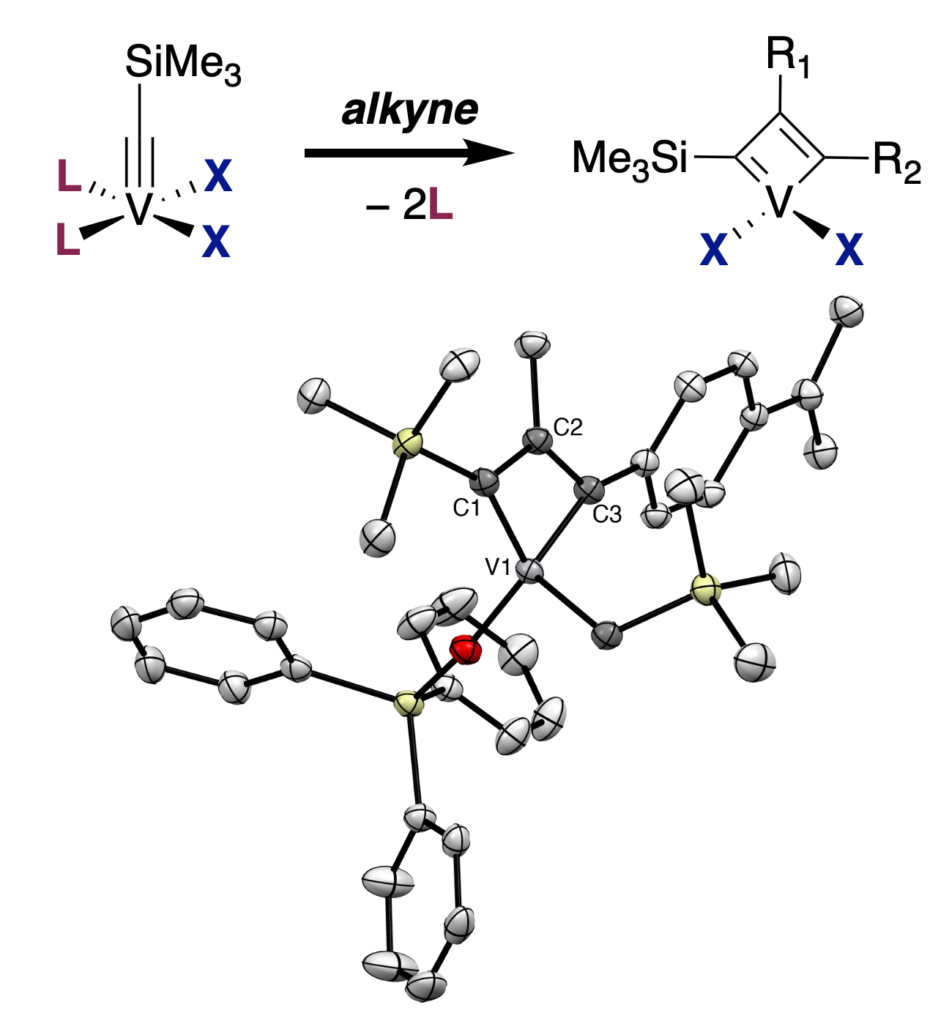
43. S. Hernandez, V. Krivovicheva, A. Sousa-Silva, Xavier Solans-Monfort, and K. V. Bukhryakov,* Toward Vanadium-Mediated Alkyne Metathesis, Chem. Sci., 2026, 17, 9132 – 9137. DOI: 10.1039/d6sc01683a
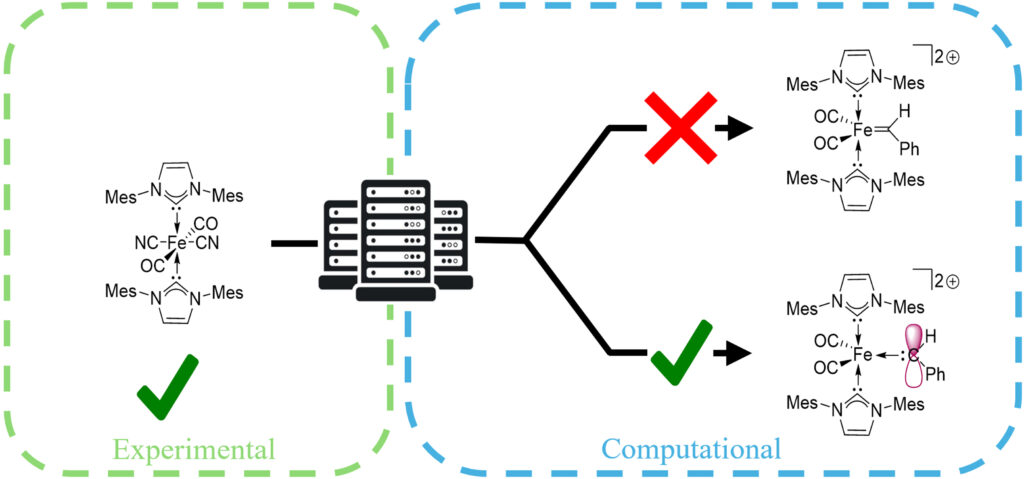
42. C. Acosta, L. Rodriguez-Santiago,V. Krivovicheva,X. Solans-Monfort,* and K. V. Bukhryakov,* Synthesis of Low-Spin Fe(II) NHC Complexes and Their Computational Evaluations in the Context of Olefin Metathesis, Organometallics, 2025, 44, 2741–2748. DOI: 10.1021/acs.organomet.5c00281
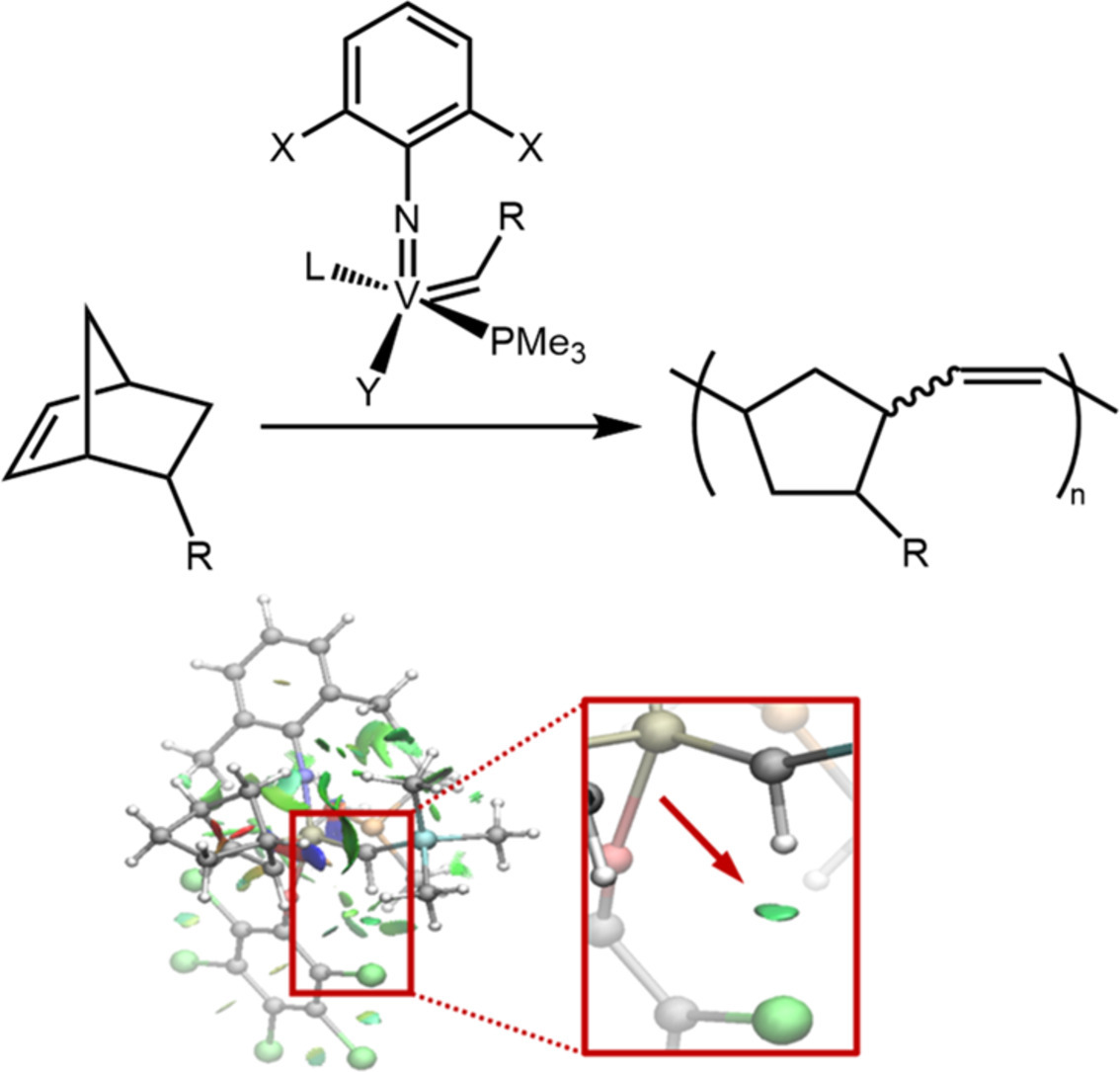
41. S. Aguero, V. Krivovicheva, M. R. Gangireddy, and K. V. Bukhryakov,* Evaluation of Steric and Electronic Parameters of NHCs on Vanadium Imido Alkylidenes for Olefin Metathesis, ACS Catal., 2025, 15, 18326–18332. DOI: 10.1021/acscatal.5c05494
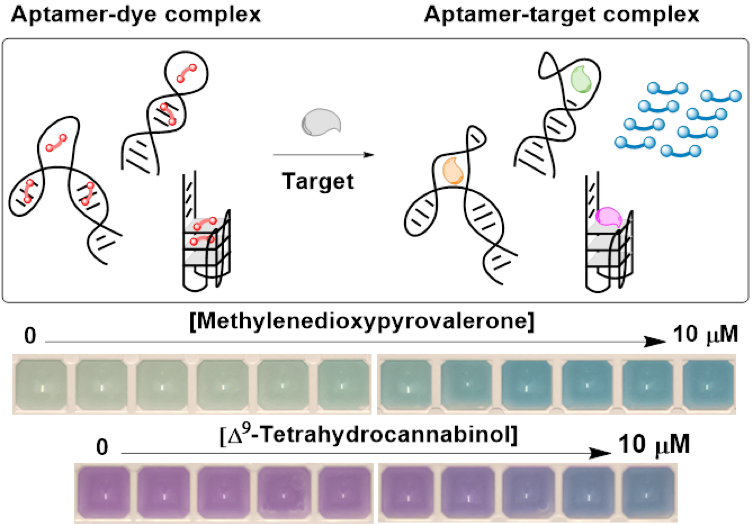
40. T. Nguyen, O. Alkhamis, A. K. Bacon, K. V. Bukhryakov, N. R. Vinueza, and Y. Xiao,* High-Contrast Aptamer-Based Merocyanine Displacement Assays for Sensitive Small Molecule Detection, ACS Sens., 2025, 10, 7799–7809. DOI: 10.1021/acssensors.5c02194
39. R. K. Haridasan, S. Hernandez, S. Kuenzig, G. Tejeda, K. V. Bukhryakov,* K. Searles,* Silica-Supported Vanadium-Oxo-Alkylidene for Self-Metathesis of Propene, J. Am. Chem. Soc., 2025, 147, 36039–36043. DOI: 10.1021/jacs.5c07854
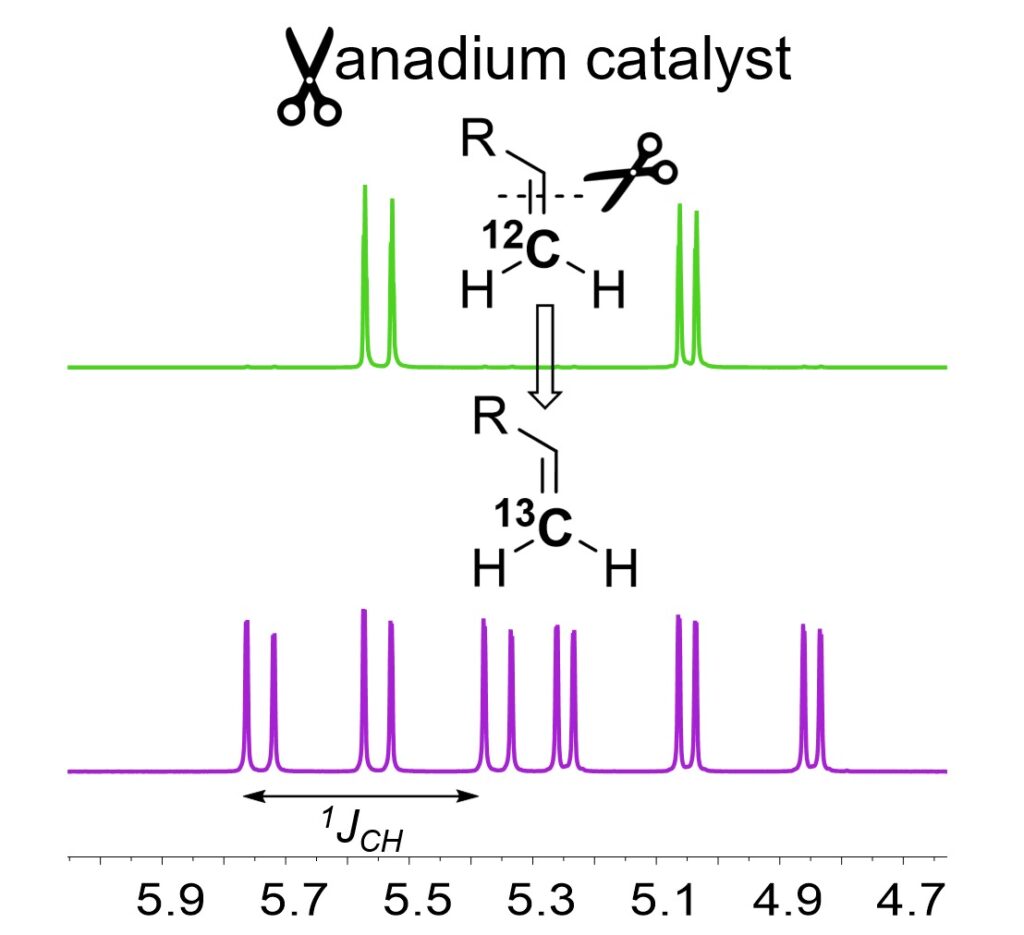
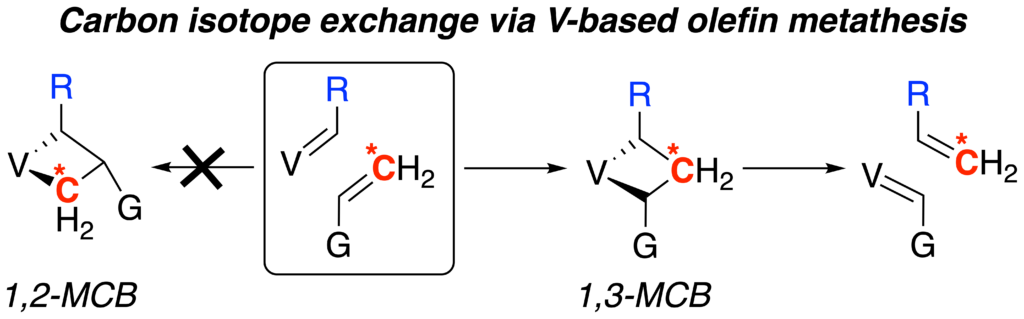
38. V. Krivovicheva, M. R. Gangireddy, A. Liu, L. B. Maya, A. M. Mebel, and K. V. Bukhryakov,* Vanadium-Mediated Carbon Isotope Exchange of Terminal Alkenes, J. Am. Chem. Soc., 2025, 147, 20212–20217. DOI: 10.1021/jacs.5c05880
37. S. Hernandez, D. S. Belov, V. Krivovicheva, S. Senthil, and K. V. Bukhryakov,* Decreasing the Bond Order Between Vanadium and Oxo Ligand to Form 3d Schrock Carbynes, J. Am. Chem. Soc., 2024, 146, 18905–18909. DOI: 10.1021/jacs.4c07588
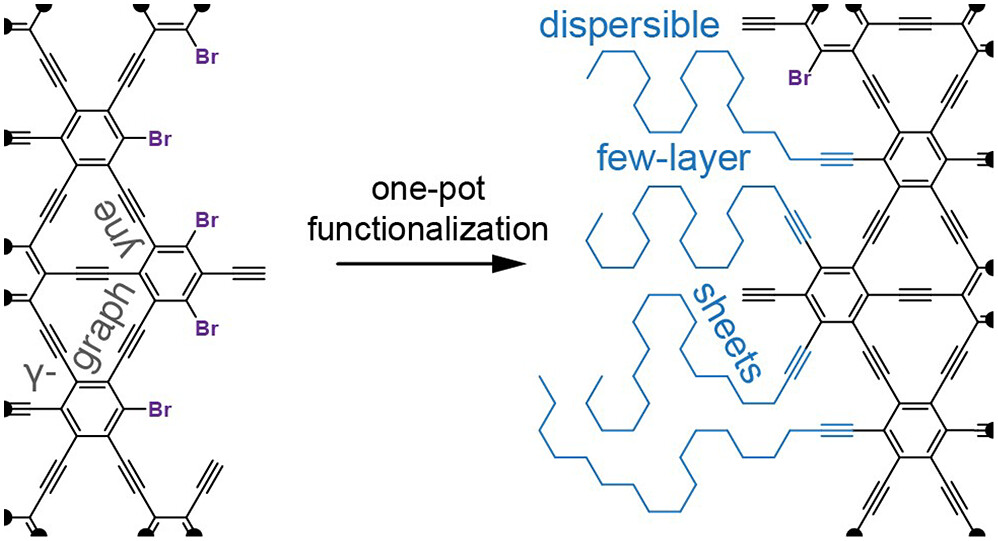
36. C. M. B. Bolding, T. K. Haraniya, G. L. Parker, W. B. Martin, V. G. Desyatkin, L. Heck, K. V. Bukhryakov, V. O. Rodionov,* Edge Functionalization of Bulk γ-Graphyne Facilitates Mechanical Exfoliation and Modulates the Mode of Sheet Stacking, J. Am. Chem. Soc., 2024, 146, 12889−12894. DOI: 10.1021/jacs.4c02064
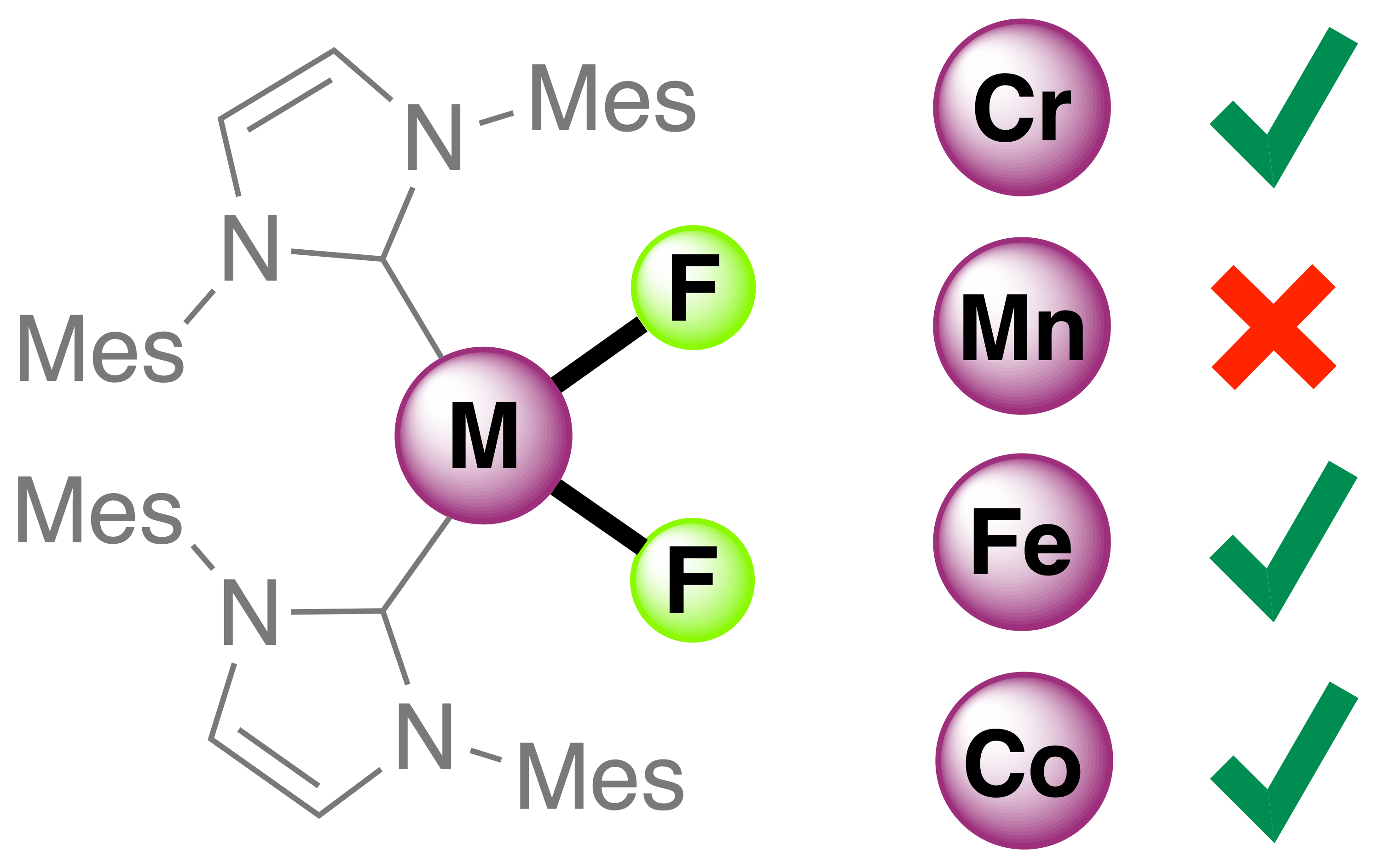
35. C. M. Acosta, D. S. Belov, A. H. Lamur, C. L. Brantley, X. Solans-Monfort, K. L. Rue, G. Christou, and K. V. Bukhryakov,* Mononuclear Four-Coordinate Bis-Fluoride Bis-NHC Complexes of Chromium(II), Iron(II), and Cobalt(II), Inorg. Chem., 2023, 62, 18108–18115. DOI: 10.1021/acs.inorgchem.3c02442
34. W. S. Farrell,* G. Tejeda, X. Solans-Monfort, É. Sá, K. V. Bukhryakov, Ring-Opening Metathesis Polymerization with Vanadium(V) Alkylidenes: Survey of Initiators, Density-Functional Theory Calculations, and Functional Group Tolerance, J. Organomet. Chem., 2023, 996, 122753. DOI: 10.1016/j.jorganchem.2023.122753
33. D. S. Belov, C. Acosta, M. Garcia-Molina K. L. Rue, X. Solans-Monfort, and K. V. Bukhryakov,* Synthesis and Activity of Vanadium Oxo NHC Alkylidenes. Remarkable Preference for Degenerate Metathesis and Application for Carbon Isotope Exchange, Organometallics, 2022, 41, 2897–2902. DOI: 10.1021/acs.organomet.2c00465
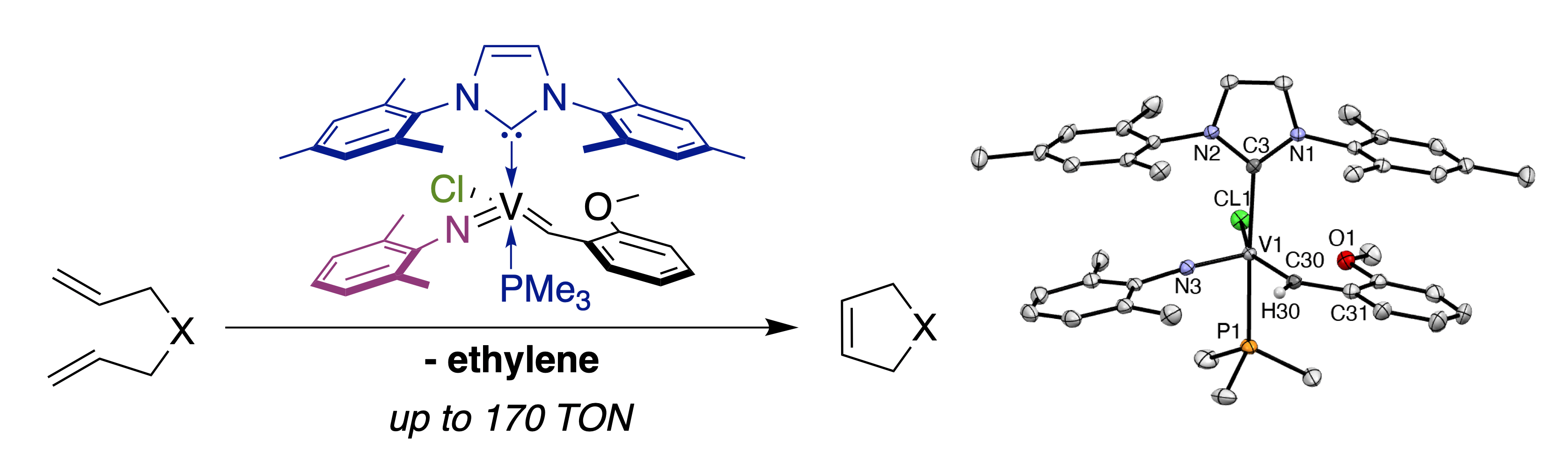
32. G. Tejeda, D. S. Belov, D. A. Fenoll, K. L. Rue, C. Tsay, X. Solans-Monfort, and K. V. Bukhryakov,* Vanadium Imido NHC Complexes for the Ring-Closing Olefin Metathesis Reactions, Organometallics, 2022, 41, 361-365. DOI: 10.1021/acs.organomet.2c00013
31. O. Alkhamis, J. Canoura, K. V. Bukhryakov, A. Tarifa, A. P. DeCaprio, and Y. Xiao,* DNA Aptamer-Cyanine Complexes as Generic Colorimetric Small-Molecule Sensors, Angew. Chem., Int. Ed., 2021, 60, 2-12. DOI: 10.1002/anie.202112305
30. D. S. Belov, D. A. Fenoll, I. Chakraborty, X. Solans-Monfort, and K. V. Bukhryakov,* Synthesis of Vanadium Oxo Alkylidene Complex and its Reactivity in Ring-Closing Olefin Metathesis Reactions, Organometallics, 2021, 40, 2939–2944. DOI: 10.1021/acs.organomet.1c00425
29. D. S. Belov, G. Tejeda, and K. V. Bukhryakov,* Olefin Metathesis by First-Row Transition Metals (invited review), ChemPlusChem, 2021, 86, 924-937. DOI: 10.1002/cplu.202100192
28. D. S. Belov, G. Tejeda, C. Tsay, and K. V. Bukhryakov,* Ring-Closing Olefin Metathesis Catalyzed by Well-Defined Vanadium Alkylidene Complexes, Chem. Eur. J, 2021, 27, 4578-4582. DOI: 10.1002/chem.202005438
27. D. S. Belov, L. Mathivathanan, M. J. Beazley, W. B. Martin, and K. V. Bukhryakov,* Stereospecific Ring-Opening Metathesis Polymerization of Norbornene Catalyzed by Iron Complexes, Angew. Chem., Int. Ed., 2021, 60, 2934-2938. DOI: 10.1002/anie.202011150 (highlighted in ChemistryViews).
26. S. Chuprun, C. M. Acosta, L. Mathivathanan, and K. V. Bukhryakov,* Molybdenum Benzylidyne Complexes for Olefin Metathesis Reactions, Organometallics, 2020, 39, 3453-3457. DOI: 10.1021/acs.organomet.0c00491
Before FIU
Postdoctoral research (MIT):
25. F. Zhai, K. V. Bukhryakov, R. R. Schrock, A. Hoveyda, C. Tsay, and P. Müller, Syntheses of Molybdenum Охо Benzylidene Complexes, J. Am. Chem. Soc., 2018, 140, 13609-13613. DOI: 10.1021/jacs.8b09616
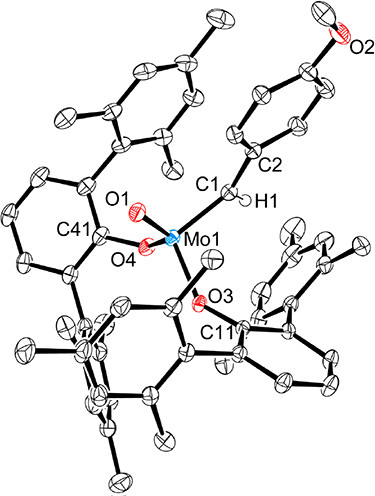
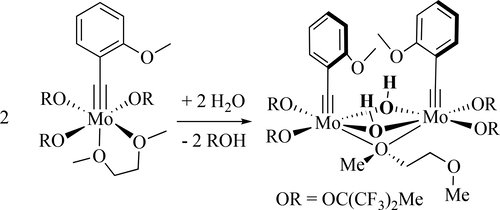
24. K. V. Bukhryakov, R. R. Schrock, A. Hoveyda, C. Tsay, and P. Müller, Syntheses of Molybdenum Охо Alkylidene Complexes Through Addition of Water to an Alkylidyne Complex, J. Am. Chem. Soc., 2018, 140, 2797-2800. DOI: 10.1021/jacs.8b00499
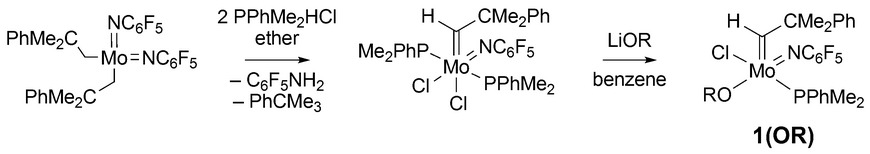
23. K. V. Bukhryakov, S. VenkatRamani, C. Tsay, A. Hoveyda, and R. R. Schrock, Syntheses of Molybdenum Adamantylimido and t-Butylimido Alkylidene Chloride Complexes Using HCl and Diphenylmethylphosphine, Organometallics, 2017, 36, 4208-4214. DOI: 10.1021/acs.organomet.7b00647
22. P. E. Sues, K. V. Bukhryakov, and R. R. Schrock, Evaluation of Several Molybdenum and Ruthenium Catalysts for the Metathesis Homocoupling of 3-Methyl-1-Butene, Helv. Chim. Acta, 2017, 100, e1700181. DOI: 10.1002/hlca.201700181
21. K. V. Bukhryakov, R. R. Schrock, A. Hoveyda, P. Müller, and J. Becker, Synthesis of 2,6-Hexatertiarybutylterphenyl Derivatives, 2,6-(2,4,6-t-Bu3C6H2)2C6H3X, where X = I, Li, OH, SH, N3, or NH2, Org. Lett., 2017, 19, 2607-2609. DOI: 10.1021/acs.orglett.7b01062
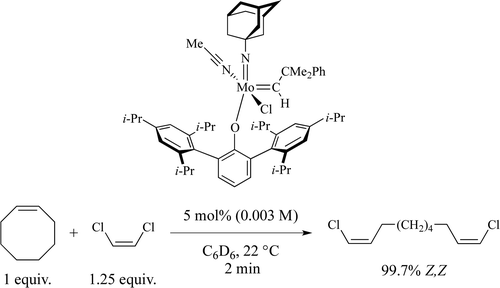
20. J. K. Lam, C. Zhu, K. V. Bukhryakov, P. Müller, A. Hoveyda, and R. R. Schrock, Synthesis and Evaluation of Molybdenum and Tungsten Monoaryloxide Halide Alkylidene Complexes for Z-Selective Cross-Metathesis of Cyclooctene and Z-1,2- Dichloroethylene, J. Am. Chem. Soc., 2016, 138, 15774-15783. DOI: 10.1021/jacs.6b10499
19. P. E. Sues, J. M. John, K. V. Bukhryakov, R. R. Schrock, and P. Müller, Molybdenum and Tungsten Alkylidene Complexes That Contain a 2-Pyridyl-substituted Phenoxide Ligand, Organometallics, 2016, 35, 3587-3593. DOI: 10.1021/acs.organomet.6b00644
Postdoctoral research (KAUST):
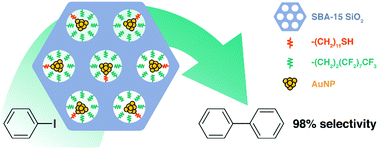
18. T. Chen, B. Chen, K. V. Bukhryakov, and V. O. Rodionov, Thiols Make for Better Catalyst: Au Nanoparticles Supported on Functional SBA-15 for Catalysis of Ullmann-type Homocouplings, Chem. Commun., 2017, 53, 11638-11641. DOI: 10.1039/C7CC06146C
17. K. B. Vu, T. Chen, S. Almahdali, K. V. Bukhryakov, and V. O. Rodionov, Hollow Nanospheres with Fluorous Interiors for Transport of Molecular Oxygen in Water, ChemistrySelect, 2016, 1, 3306-3309. DOI: 10.1002/slct.201600602
16. K. V. Bukhryakov, V. G. Desyatkin, and V. O. Rodionov, Cooperative Organocatalysis of Mukaiyama–Type Aldol Reactions by Thioureas and Nitro Compounds, Chem. Commun., 2016, 52, 7576-7579. DOI: 10.1039/C6CC01984F
15. C. Mugemana, K. V. Bukhryakov, O. Bertrand, K. B. Vu, J.-F. Gohy, N. Hadjichristidis, V. O. Rodionov, Ring opening metathesis polymerization of cyclopentene using a ruthenium catalyst confined by a branched polymer architecture, Polym. Chem., 2016, 7, 2923-2928. DOI: 10.1039/C6PY00389C
14. K. V. Bukhryakov, C. Mugemana, K. B. Vu, V. O. Rodionov, Palladium-N-Heterocyclic Carbene Pre-Catalyst Site-Isolated in the Core of a Star Polymer, Org. Lett., 2015, 17, 4826-4829 DOI: 10.1021/acs.orglett.5b02388 (selected by the Editorial Board of Synfacts for its important insights, Synfacts, 2016, 12, 99 DOI: 10.1055/s-0035-1561082).
13. K. B. Vu, K. V. Bukhryakov, D. H. Anjum, V. O. Rodionov, Surface-Bound Ligands Modulate Chemoselectivity and Activity of a Bimetallic Nanoparticle Catalyst, ACS Catal., 2015, 5, 2529-2533 (K. B. Vu and K. V. Bukhryakov contributed equally). DOI: 10.1021/acscatal.5b00262
12. K. V. Bukhryakov, S. Almahdali, and V. O. Rodionov, Amplification of Chirality through Self-Replication of Micellar Aggregates in Water, Langmuir, 2015, 31, 2931-2935. DOI: 10.1021/la504984j
11. B. Chen, K. V. Bukhryakov, R. Sougrat, and V. Rodionov, An Enzyme-Inspired Functional Surfactant for Aerobic Oxidation of Activated Alcohols to Aldehydes in Water, ACS Catal., 2015, 5, 1313-1317. DOI: 10.1021/cs5020018
10. K. V. Bukhryakov, V. G. Desyatkin, J. P. O’Shea, S. R. Almahdali, V. Solovyeva, and V. Rodionov, Cooperative Catalysis With Block Copolymer Micelles: A Combinatorial Approach, ACS Comb. Sci., 2015, 17, 76-80 DOI: 10.1021/co5001713 (selected as ACS Editors’ Choice article, Jan. 13, 2015 and highlighted in C&EN, Jan. 19, 2015)
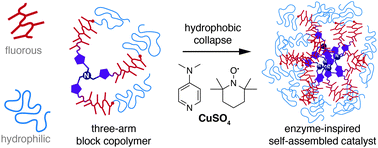
9. C. Mugemana, B. Chen, K. V. Bukhryakov, and V. Rodionov, Star Block-Copolymers: Enzyme-Inspired Catalysts for Oxidation of Alcohols in Water, Chem. Commun., 2014, 50, 7862-7865. DOI: 10.1039/C4CC03370A
Ph.D. research (MSU and CDI):
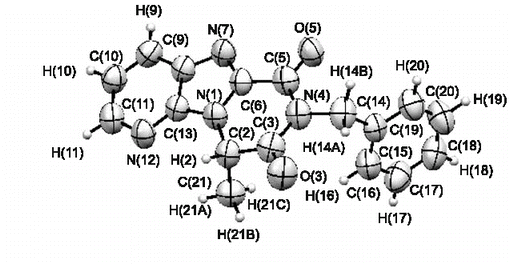
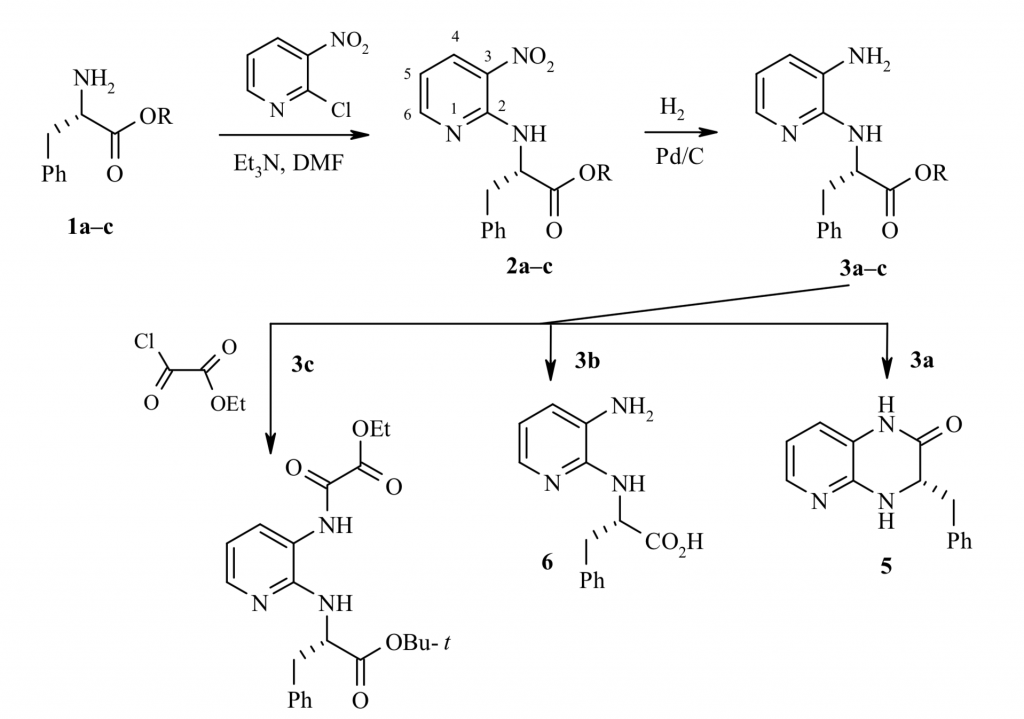
8. K. V. Bukhryakov, A. V. Kurkin, M. A. Yurovskaya, Synthesis of Imidazo[4,5-b]pyridines with a Chiral Substituent at the Nitrogen Atom and their Conversion to Piperazine Derivatives, Chem. Heterocycl. Compd. (N.Y.), 2012, 48, 773-784. DOI: 10.1007/s10593-012-1056-5
7. K. V. Bukhryakov, A. V. Kurkin, and M. A. Yurovskaya, Synthetic Approaches to Imidazo[4,5-b]pyridine Derivatives (review), Chem. Heterocycl. Compd. (N.Y.), 2011, 47, 533-557. DOI: 10.1007/s10593-011-0797-x
6. A. V. Kurkin, K. V. Bukhryakov, M. A. Yurovskaya, Synthesis of 1,2,3,4-tetrahydro[2,3-b]pyrazindiones with a Chiral Substituent at the Nitrogen, Chem. Heterocycl. Compd. (N.Y.), 2009, 45, 188-193. DOI: 10.1007/s10593-009-0249-z
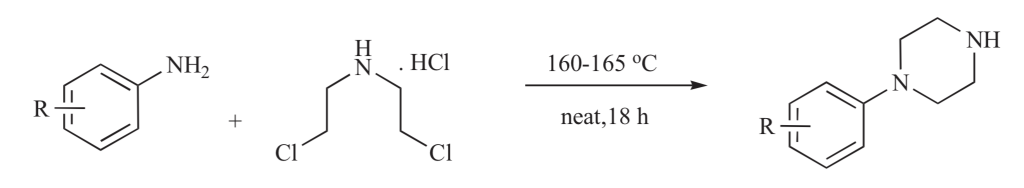
5. I. Konstantinov, K. Bukhryakov, Y. Gezentsvey, and M. Krasavin, Practical Method for Parallel Synthesis of Diversely Substituted 1-Phenylpiperazines, Lett. Org. Chem., 2011, 8, 628-630. DOI: 10.2174/157017811799304386
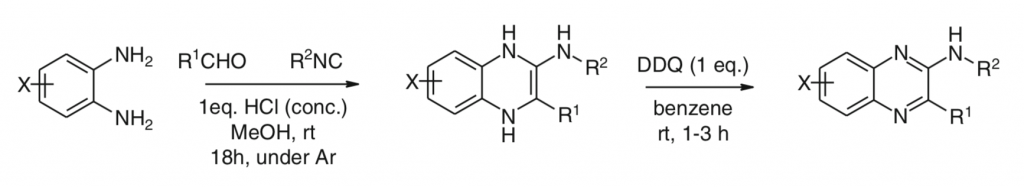
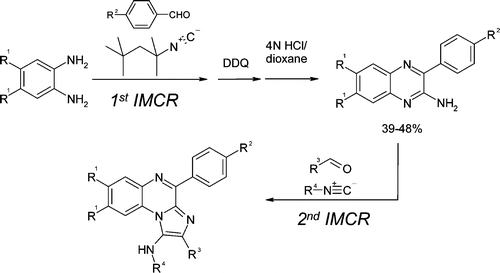
4. M. Krasavin, V. Parchinsky, S. Shkavrov, and K. Bukhryakov, Applications of Quinoxaline Synthesis via Isocyanide-Based MCR. Multi-Component Reactions in Drug Discovery, Adv. Exp. Med. Bio, 2011, 699, 88-89. DOI: 10.1007/978-1-4419-7270-5_3
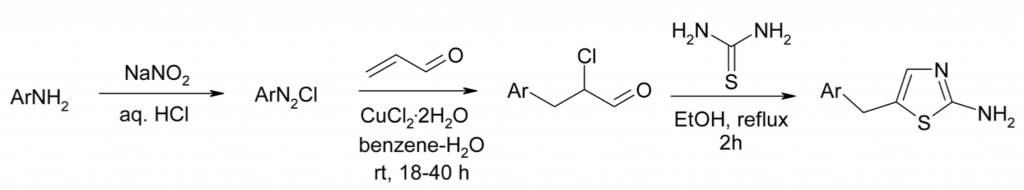
3. M. Krasavin, R. Karapetian, I. Konstantinov, Y. Gezentsvey, K. Bukhryakov, E. Godovykh, O. Soldatkina, Y. Lavrovsky, A.V. Sosnov, A.A. Gakh, Discovery and Potency Optimization of 2-Amino-5-arylmethyl-1,3-thiazole Derivatives as Potential Therapeutic Agents for Prostate Cancer, Arch. Pharm., 2009, 342, 420-427. DOI: 10.1002/ardp.200800201
2. M. Krasavin, S. Shkavrov, V. Parchinsky, and K. Bukhryakov, Imidazo[1,2-a]quinoxalines Accessed via Two Sequential Isocyanide-Based Multicomponent Reactions, J. Org. Chem., 2009, 74, 2627-2629. DOI: 10.1021/jo900050
1.M. Krasavin, S. Tsirulnikov, M. Nikulnikov, Y. Sandulenko, and K. Bukhryakov, tert-Butyl Isocyanide Revisited as a Convertible Reagent in the Groebke–Blackburn Reaction, Tetrahedron Lett., 2008, 49, 7318-7321. DOI: 10.1016/j.tetlet.2008.10.046